The Intriguing Bonds Of DNA: Understanding Hydrogen Bonds In The Double Helix
At the heart of all living organisms lies a remarkable molecule known as DNA, or deoxyribonucleic acid. This complex structure carries the genetic blueprints of life and is fundamental to the functioning of cells. One of the most fascinating aspects of DNA is its double helix shape, which is maintained by the interactions between its two strands. More specifically, the two strands of a DNA molecule are held together by hydrogen bonds, a detail that may seem small but is crucial to the stability and integrity of genetic information. In this article, we will delve into the nature of these hydrogen bonds, exploring how they contribute to the structure and function of DNA. We will also discuss the implications of these bonds in biological processes and why they are essential for life as we know it.
Understanding the role of hydrogen bonds in DNA not only provides insight into molecular biology but also opens the door to advancements in genetic research and biotechnology. As scientists continue to unravel the complexities of DNA, the significance of these bonds becomes even more apparent. By examining the two strands of a DNA molecule and their interactions, we can appreciate the elegance and precision of life at a molecular level.
Join us as we explore this captivating topic and answer some of the most pressing questions about the two strands of a DNA molecule, the hydrogen bonds that connect them, and their implications for life on Earth.
What Are Hydrogen Bonds?
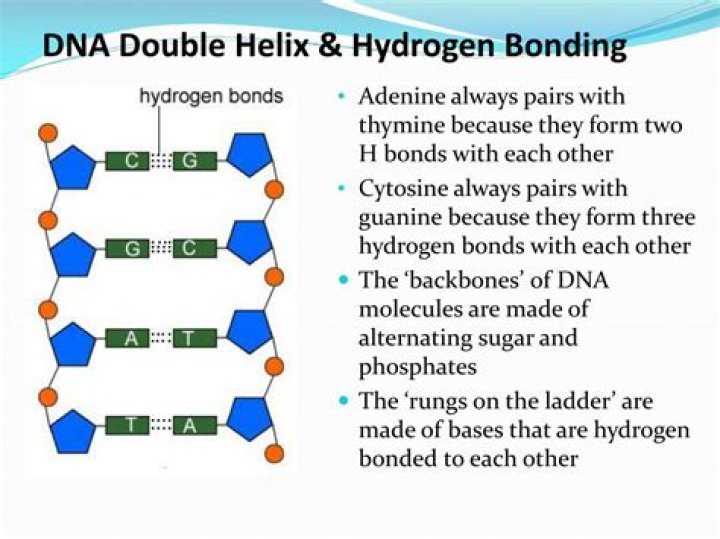
Hydrogen bonds are weak interactions that occur between molecules when hydrogen is covalently bonded to a highly electronegative atom such as oxygen or nitrogen. In the context of DNA, hydrogen bonds form between the nitrogenous bases of the two strands. This bonding is crucial for the stability of the double helix structure.
How Do Hydrogen Bonds Contribute to DNA Structure?
The two strands of a DNA molecule are held together by hydrogen bonds between complementary base pairs. Each base pair consists of two nucleobases: adenine (A) pairs with thymine (T), and guanine (G) pairs with cytosine (C). The specific pairing is due to the formation of two hydrogen bonds between A and T and three hydrogen bonds between G and C, ensuring the precise alignment of the strands.
Why Are Hydrogen Bonds Important for DNA Function?
Hydrogen bonds play a vital role in the replication and transcription of DNA. During replication, the strands must separate, and the hydrogen bonds must break. This allows each strand to serve as a template for creating new complementary strands. Additionally, during transcription, hydrogen bonds facilitate the process of copying a gene into RNA.
What Happens If Hydrogen Bonds Are Disrupted?
Disruption of hydrogen bonds can lead to significant consequences for an organism. If the hydrogen bonds are weakened or broken, it can result in mutations, which may affect protein synthesis or lead to diseases such as cancer. Understanding these implications highlights the importance of the two strands of a DNA molecule being held together by hydrogen bonds for maintaining genetic integrity.
How Do Environmental Factors Affect Hydrogen Bonds in DNA?
Environmental factors such as temperature, pH, and chemical exposure can influence the stability of hydrogen bonds in DNA. For instance, increased temperatures can lead to denaturation, where the hydrogen bonds break, and the double helix unwinds. This is a critical consideration in laboratory settings and when studying the effects of various substances on genetic material.
Can Hydrogen Bonds Be Utilized in Biotechnology?
The understanding of hydrogen bonds has paved the way for numerous advancements in biotechnology. For example, techniques like polymerase chain reaction (PCR) rely on the principles of hydrogen bonding to amplify specific DNA sequences. This technology has revolutionized fields such as forensic science, medical diagnostics, and genetic research.
Conclusion: The Significance of Hydrogen Bonds in DNA
In conclusion, the two strands of a DNA molecule are held together by hydrogen bonds, which play a fundamental role in the structure, function, and stability of DNA. These bonds not only ensure the accurate replication and transcription of genetic information but also have significant implications for the health and functioning of living organisms. As we continue to explore the intricacies of DNA, the importance of hydrogen bonds remains a key focus in molecular biology, genetics, and biotechnology. Understanding these bonds furthers our comprehension of life itself and opens new avenues for scientific discovery.
Unraveling The Mystery: Steve Harvey Desth
Exploring The Rocky Age: A Journey Through Time
Unveiling The Truth: Donald Trump Jr. Height And More
The two strands of DNA are held together by bonds of(a) Nitrogen(b

Which Pair Of Nitrogenous Bases Will Form A Bond In A Dna Molecule
Question Video Identifying the Type of Bonds that Form Between DNA


